Treatment with nazartinib (EGF816), a third generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI), provided durable responses in adult patients with EGFR-mutated non-small lung cancer (NSCLC) who had not received prior treatment.
The primary efficacy and safety data from a phase II trial of single agent nazartinib were presented at the ESMO 2018 Congress in Munich, Germany.
Nazartinib selectively targets L858R and ex19del activating mutations, as well as T790M resistance mutants, while sparing wild type EGFR, explained Daniel SW Tan, Medical Oncology, National Cancer Centre Singapore, Singapore.
This phase I/II multicentre study (NCT02108964; EUDRACT number 2013-004482-14) of nazartinib was conducted in treatment-naive patients with advanced EGFR-mutant NSCLC harbouring activating EGFR L858R and/or ex19del mutations.
All 45 patients received the recommended phase II oral dose of 150 mg once daily on a continuous schedule.
Anti-tumour activity, including overall response rate (ORR) per RECIST v1.1, as assessed by blinded independent central review (BICR), served as the primary objective, and secondary objectives included safety, tolerability, and pharmacokinetics.
The median age was 64 years, 60% of patients in the trial were female, and 62% were Asian. Fifty-eight percent had ECOG performance status 1 and 18 (45%) patients had brain metastasis at baseline. EGFR mutations were ex19del in 56% of patients, L858R in 40%, and 4% of patients had other EGFR mutations.
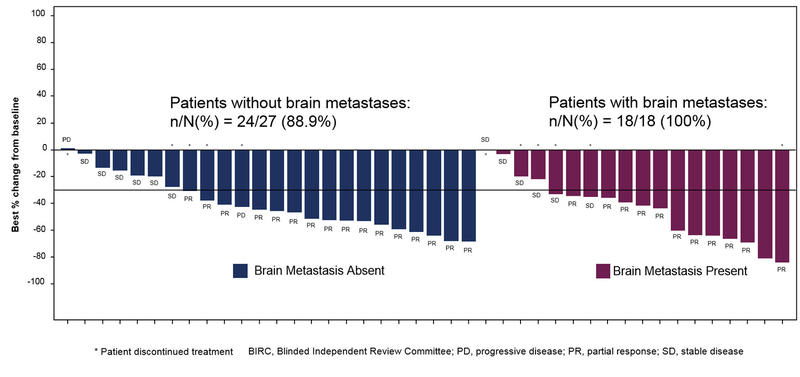
High response and disease control rates observed with nazartinib
Twenty-nine of 45 patients demonstrated a response to nazartinib, yielding an ORR of 64% (95% confidence interval [CI], 49% - 78%). One patient achieved complete response.
At data cutoff on 22 March 2018, responses were ongoing in 27 of the 29 responding patients. The 6-month duration of response rate (DoR) was 91%, and the median DoR was not estimable (NE). The disease control rate was 93%.
The 6-month progression-free survival rate was 83% (median NE) and the 6-month overall survival rate was 95% (median NE) with nazartinib.
Evaluation of the 17 patients with baseline brain metastasis in non-target lesions showed 9 (53%) patients had resolution of brain metastasis. One of the 27 patients without baseline brain metastasis developed a new brain metastasis on study.
Nazartinib was well-tolerated
The 45 patients had a median duration of nazartinib exposure of 43.3 weeks.
Frequently reported adverse events (AEs; any grade ≥ 20%) regardless of causality included diarrhea (38%), maculopapular rash (31%), stomatitis (24%), cough (22%), decreased appetite (22%), pruritus (20%), and pyrexia (20%).
The most frequently occurring grade 3 to 4 AE (≥ 5%) was maculopapular rash, which was reported in 9% of the patients.
Nazartinib therapy was discontinued by 9 patients due to progressive disease, one patient discontinued due to an adverse event, maculopapular rash, one due to patient choice, and one discontinuation was due to death.
Discussant Points
Dr Raffaele Califano of the Department of Medical Oncology, The Christie and Manchester University Hospital, Manchester, UK who discussed the study findings said that they point to an effective third generation EGFR-TKI with good brain penetration. The PFS, DoR and OS are not mature yet and there is a higher incidence of grade ≥3 rash than with osimertinib. However, Dr Raffaele Califano questioned if we need another third generation EGFR-TKI.
Conclusions
The investigators concluded that nazartinib demonstrated promising efficacy, and provided durable responses in treatment-naive patients with advanced EGFR-mutant NSCLC, including patients with baseline brain metastases.
Nazartinib also demonstrated a tolerable safety profile.
Disclosure
This trial was sponsored by Novartis.
Reference
LBA61 – Tan DS, Kim SW, Sequist LV, et al. Phase II results for single-agent nazartinib (EGF816) in adult patients (pts) with treatment-naive EGFR-mutant non-small cell lung cancer (NSCLC).