A group of researchers from the University of California, San Francisco, USA reported in the Nature Medicine that pharmacologic inhibition of fatty acid oxidation (FAO) decreased energy metabolism in MYC-driven transgenic triple negative breast cancer (TNBC) model and in a MYC-overexpressing TNBC patient–derived xenograft (PDX). They suggest that inhibition of FAO as a potential therapeutic strategy for this subset of breast cancer should be further investigated.
Expression of the oncogenic transcription factor MYC is disproportionately elevated in TNBC, as compared to estrogen receptor–, progesterone receptor– or human epidermal growth factor 2 receptor–positive breast cancer. The researchers have shown that MYC alters metabolism during tumourigenesis. However, the role of MYC in TNBC metabolism remained mostly unexplored.
The study investigators hypothesized that MYC-dependent metabolic dysregulation is essential for the growth of MYC-overexpressing TNBC cells and may identify new therapeutic targets for this clinically challenging subset of breast cancer.
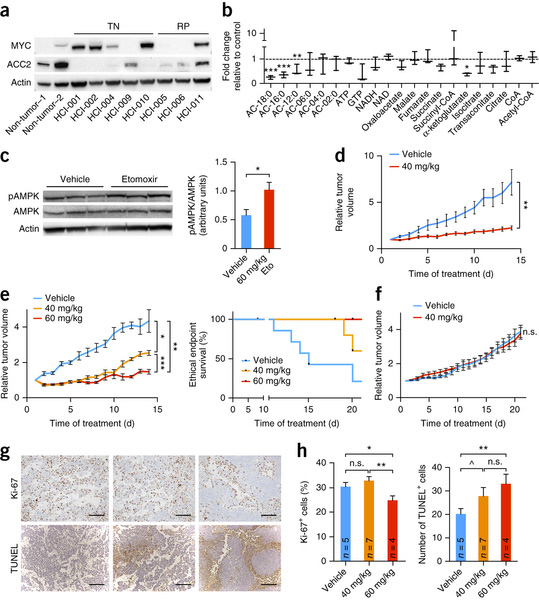
To test this hypothesis, they investigated tumour metabolism in a transgenic model of MYC-overexpressing TNBC. They used mass spectrometry and metabolite analysis revealed a number of dysregulated pathways that are commonly associated with tumourigenesis, including glycolysis, the tricarboxylic acid cycle and fatty acid metabolism. Although fatty acid synthesis is upregulated in many types of cancer, a MYC-dependent role for FAO in breast tumourigenesis has not previously been described. Considering the proximity of primary breast tumours to the adipose-rich mammary gland, the investigators chose to focus on the dysregulation of FAO in MYC-overexpressing TNBC cells.
By using a targeted metabolomics approach, they identified FAO intermediates as being dramatically upregulated in a MYC-driven model of TNBC. They also identified a lipid metabolism gene signature in patients with TNBC that were identified from The Cancer Genome Atlas database and from multiple other clinical data sets, implicating FAO as a dysregulated pathway that is critical for TNBC cell metabolism.
The researchers found that pharmacologic inhibition of FAO decreased energy metabolism in MYC-overexpressing TNBC cells and blocked tumour growth in a MYC-driven transgenic TNBC model and in a MYC-overexpressing TNBC PDX. These findings demonstrate that MYC-overexpressing TNBC shows an increased bioenergetic reliance on FAO.
Elevated MYC expression was recently discovered to be a defining factor of TNBC. The present study is among the first to investigate the role of MYC in TNBC metabolism in vivo. FAO is upregulated in MYC-overexpressing TNBC cells and TNBC is sensitive to FAO inhibition in a MYC-dependent manner. This work supports a critical role for FAO in TNBC, and it identifies high levels of MYC expression as a marker for this dependence. On the basis of study findings, the authors concluded that inhibition of FAO should be further investigated as a therapeutic strategy for MYC-overexpressing TNBC.