An update of results of the CheckMate 067 trial (NCT01844505) at 5 years presented at the ESMO Congress 2019 in Barcelona, Spain showed that both nivolumab in combination with ipilimumab and nivolumab alone provided significant improvements in overall survival (OS), progression-free survival (PFS), and objective response rate (ORR) over ipilimumab in patients with advanced treatment-naive melanoma.
On behalf of colleagues, James Larkin of the Royal Marsden Hospital NHS Foundation Trust in London, United Kingdom presented findings from the phase III CheckMate 067 trial in advanced melanoma after a minimum follow-up of 60 months.
The study randomised 945 treatment-naive patients with stage III or IV melanoma; 314 patients received nivolumab at 1 mg/kg plus ipilimumab at 3 mg/kg every 3 weeks for 4 doses followed by nivolumab at 3 mg/kg every 2 weeks, 316 patients received nivolumab at 3 mg/kg every 2 weeks plus ipilimumab-matched placebo, and 315 patients received ipilimumab at 3 mg/kg every 3 weeks for 4 doses plus nivolumab-matched placebo until progression or unacceptable toxicity occurred. The patients were stratified by tumour programmed death ligand 1 (PD-L1) status, tumour BRAF mutation status, and metastasis stage.
The co-primary endpoints were OS and PFS. Since the study was not designed to compare the nivolumab treatment arms, descriptive analyses were performed to evaluate efficacy between combined nivolumab plus ipilimumab and nivolumab monotherapy, as well as treatment-free status and health-related quality of life (HRQoL) assessed using the EQ-5D-3L utility index.
Improved outcomes were observed in both nivolumab-containing arms compared with ipilimumab monotherapy
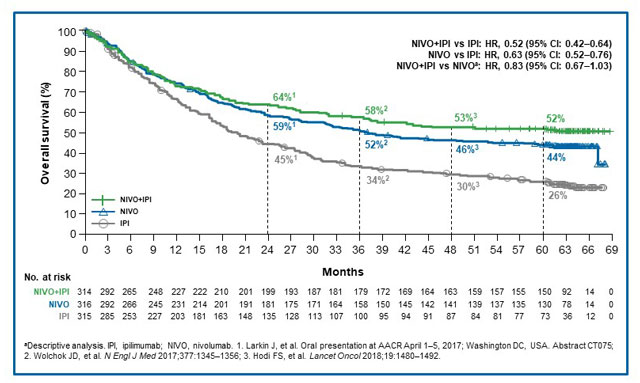
These long-term data showed that patients with melanoma treated with nivolumab, delivered either as monotherapy or in combination with ipilimumab, continued to show superior OS, PFS, and response rates compared with those on ipilimumab.
With a minimum of 60 months of follow-up, median OS in the nivolumab plus ipilimumab, nivolumab, and ipilimumab arms was >60 months (not yet reached [NR]; 95% confidence interval [CI], 38.2–NR), 36.9 months (95% CI, 28.2–58.7), and 19.9 months (95% CI, 16.8–24.6), respectively; 5-year OS rates were 52% (95% CI, 46–57), 44% (95% CI, 39–50), and 26% (95% CI, 22–31).
Median PFS was 11.5 months (95% CI, 8.7–19.3) with nivolumab plus ipilimumab, 6.9 months (95% CI, 5.1–10.2) with nivolumab, and 2.9 months (95% CI, 2.8–3.2) with ipilimumab. Five-year PFS rates were 36% (95% CI, 31–42), 29% (95% CI, 24–35), and 8% (95% CI, 5–12), respectively.
In the respective treatment arms, ORRs were 58%, 45%, and 19%. The median duration of response was NR in both nivolumab arms compared with 14.4 months in the ipilimumab arm.
The median time from randomisation to administration of subsequent systemic therapy was NR (95% CI, 59.6–NR) with nivolumab plus ipilimumab and 25.2 months (95% CI, 16.0–43.2) with nivolumab alone compared with 8.0 months (95% CI, 6.5–8.7) with ipilimumab.
At 60 months among patients who were alive and followed on study, 112/151 (74%), 75/130 (58%) and 30/67 (45%) of those treated with nivolumab plus ipilimumab, nivolumab, and ipilimumab remained free from subsequent systemic therapy, respectively.
Durable clinical benefit was demonstrated with nivolumab-based treatment across clinically relevant subgroups
OS was improved in the nivolumab-containing arms compared with the ipilimumab arm irrespective of tumour BRAF mutation status. Five-year OS rates in patients with tumour BRAF mutations were 60% with nivolumab plus ipilimumab, 46% with nivolumab, and 30% with ipilimumab; and 48%, 43%, and 25% with the respective treatments in patients with wild-type tumour BRAF.
Similarly, improved OS was observed with nivolumab-based treatment irrespective of baseline lactate dehydrogenase (LDH) levels: in patients with LDH levels at or under the upper limit of normal (ULN), 5-year OS rates with the respective treatments were 60%, 53%, and 34%; and 38%, 28%, and 15% in patients with LDH levels above the ULN.
Finally, improved OS with the respective treatments was seen in patients with tumour PD‑L1 expression ≥5%, among whom 5-year OS rates were 57%, 51%, and 33%; rates were 51%, 43%, and 24% in patients with PD-L1 expression <5%.
Discussant points
Ulrich Keilholz of the Charité Comprehensive Cancer Center, Berlin, Germany who discussed the value of CheckMate 067 update said that it is excellent study. In particular, it is very useful to have robust 5 year data. Median OS over 60 months is a great achievement for patients with metastatic melanoma. He further discussed where do we stand today in 5 year survival in comparison to other tumour types. Next set of research questions to be addressed according Prof. Keilholz are if combination treatments can improve outcome. The results with IDO inhibitors so far are negative. Multiple combination immune therapies are under investigation. Philosophy of targeted immunotherapy is still only partially developed. He questioned if the combination of checkpoint inhibitors plus MAPK inhibitors is effective.
Conclusions
The authors pointed out that this 5-year analysis represents the longest phase III follow-up for checkpoint inhibitor combination therapy to date. These findings demonstrate that long-term survival is improved with nivolumab in combination with ipilimumab or as monotherapy compared with ipilimumab.
In descriptive analyses, the combination treatment was associated with improved survival compared with nivolumab alone and a higher likelihood of being alive and treatment-free compared with nivolumab or ipilimumab alone.
The authors further noted that HRQoL was maintained during or following treatment with nivolumab plus ipilimumab or nivolumab monotherapy over 5 years of follow-up.
The study findings are simultaneously published in the NEJM.
Disclosure
This study was funded by Bristol-Myers Squibb.
References
LBA68_PR – Larkin JMG, Chiarion-Sileni V, Gonzalez R, et al. 5-year survival outcomes of the CheckMate 067 phase 3 trial of nivolumab plus ipilimumab (NIVO+IPI) combination therapy in advanced melanoma.
Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med; Advance online publication 28 September 2019. DOI: 10.1056/NEJMa1910836.