Patients with newly diagnosed advanced ovarian cancer that were treated with niraparib after achieving a response to first-line platinum-based chemotherapy had a significantly reduced risk of disease recurrence or death compared to similar patients receiving placebo, according to findings presented at the ESMO Congress 2019 in Barcelona, Spain.
Lead author Antonio González Martín, Medical Oncology, Grupo Español de Investigación en Cáncer de Ovario (GEICO) & Clinica Universidad de Navarra in Madrid, Spain, explained that this study evaluated the efficacy of niraparib following first-line chemotherapy in patients with newly diagnosed advanced ovarian cancer. The study was prompted by earlier reports that niraparib improves progression-free survival (PFS) in patients with recurrent ovarian cancer when administered after platinum-based chemotherapy.
Niraparib is a PARP inhibitor with significant activity in tumours with germline BRCA mutation that also shown activity in those with wild-type BRCA, as well as in tumours harbouring homologous recombination deficiency (HRD); thus, both studies enrolled patients regardless of BRCA status.
Professor González Martín and colleagues conducted this double-blind, placebo-controlled phase III trial (NCT02655016) of niraparib in patients that had been newly diagnosed with advanced high-grade serous or endometrioid ovarian, primary peritoneal, or fallopian tube cancer. Eligible patients had achieved complete response (CR) or partial response (PR) to first-line platinum-based chemotherapy. Stratification factors included: best response to the first-line chemotherapy (CR versus PR), receipt of neoadjuvant chemotherapy (yes versus no), and HRD status (positive versus negative versus unknown) per the Myriad myChoice HRD test.
The women were randomised to receive 300 mg oral niraparib or placebo once daily. Of the 487 patients randomly assigned to niraparib and 246 to placebo, 247 and 126 patients in the respective treatment arms were HRD-positive (approximately 50%). Neoadjuvant chemotherapy had been administered to 67% of the patients, and 31% had achieved PR following first-line chemotherapy. Stage IV disease was reported in 35% of patients.
The primary endpoint was PFS by blinded independent central review (BICR), as analysed using a stratified Cox proportional hazards model and hierarchically tested in HRD-positive patients and the overall population thereafter.
Greater niraparib response observed in HRD-positive patients
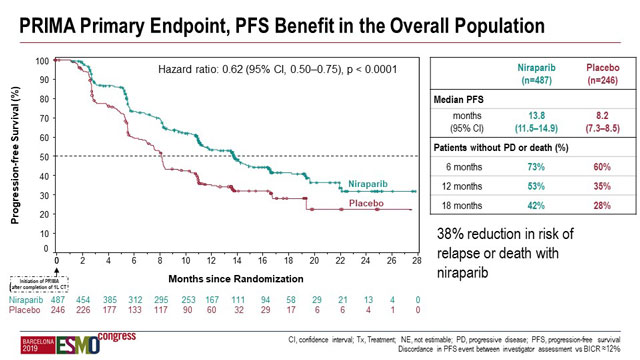
Patients in the overall population treated with niraparib demonstrated substantial improvement in PFS compared to patients on placebo that was even greater in the HRD-positive subgroup. Median PFS in the overall population was 13.8 months (95% confidence interval [CI], 11.5–14.9) with niraparib compared to 8.2 months (95% CI 7.3–8.5) with placebo (hazard ratio [HR] 0.62; 0.5–0.75; p < 0.0001).
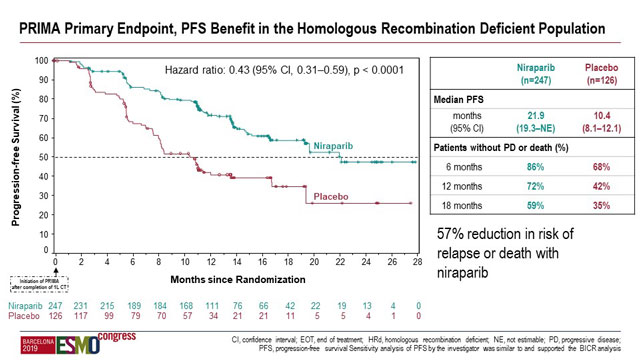
In the HRD-positive subgroup median PFS was 21.9 months (95% CI, 19.3-NR) versus 10.4 months (95% CI, 8.1-12.1) in the respective treatment arms (HR 0.43; 95% CI, 0.31–0.59; p < 0.0001).
The most commonly reported grade ≥3 adverse events were anaemia in 31% of patients, thrombocytopenia in 29%, and neutropenia in 13% of patients overall. No treatment-related deaths occurred.
Discussant points
Ana Oaknin of the Vall d´Hebron Institute of Oncology, Barcelona, Spain who discussed the study findings divided her talk in order to cover next important questions: is the benefit of adding a PARPi as maintenance therapy to first-line treatment clinically meaningful enough to justify its use as a new standard of care, can biomarker status guide in the selection of patients who benefit most, could the different control arms in the trials have modified the outcomes, can we hypothesize which sequence of therapies is the best for our patients, and are there any toxicity concerns about the use in first-line therapy. She concluded that we are witnessing a paradigm shift in the first-line treatment for advanced ovarian cancer patients.
Conclusions
The authors concluded that niraparib significantly reduced the risk of disease recurrence or death by demonstrating significantly improved PFS in patients with newly diagnosed advanced ovarian cancer. The PFS was more prolonged with niraparib versus placebo in the HRD-positive subgroup. No new safety signals were identified.
These findings suggest that niraparib be considered as a treatment option for patients with advanced ovarian following successful completion of first-line chemotherapy.
The study findings are simultaneously published in the NEJM.
Disclosure
This study was funded by TESARO: A GSK Company.
References
LBA1 – González Martín A, Pothuri B, Vergote IB, et al. Niraparib therapy in patients with newly diagnosed advanced ovarian cancer (PRIMA/ENGOT-OV26/GOG-3012 study).
González-Martín A, Pothuri B, Vergote I, et al. Niraparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N Engl J Med; Advance online publication 28 September 2019. DOI: 10.1056/NEJMoa1910962.