A potentially targetable gene alteration, NTRK fusion, which occurs rarely in Caucasian patients with lung adenocarcinoma, has been detected only in one Japanese man with lung cancer in a big genomic screening project, according to findings presented at the ESMO Asia 2017 in Singapore, an Annual Congress organised by the European Society for Medical Oncology.
Atsushi Nakamura, Department of Pulmonary Medicine, Sendai Kousei Hospital, Sendai, Japan, noted that several differences exist between East Asian and Caucasian patients with lung cancer in the frequency of actionable gene alterations, such as EGFR and KRAS mutations. Specifically, NTRK fusions have been recently identified as a therapeutic target in 3.3% of Caucasian patients with lung adenocarcinoma, but whether they occur and at what frequency in East Asian patients is unknown.
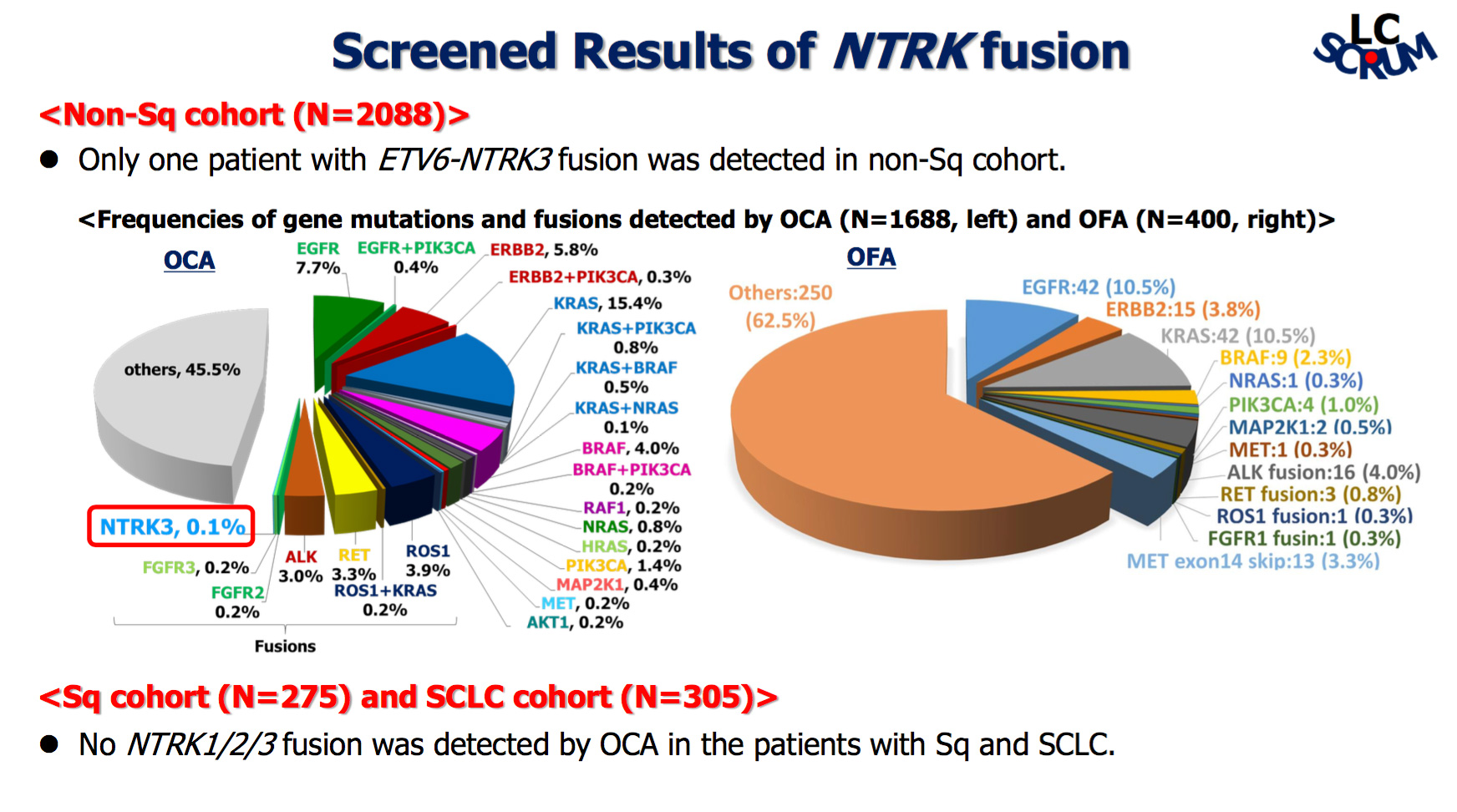
ProfessorNakamura and colleagues evaluated data collected in a nationwide lung cancer genomic screening project in Japan (LC-SCRUM-Japan) that was initiated in February 2013, with 4118 lung cancer patients enrolling by April 2017. Of these patients 2088 patients with non-squamous lung cancer, 275 with squamous, and 305 patients with small-cell lung cancer were screened using a next generation sequencing (NGS) platform, Oncomine™ Focus Assay (OFA) specific for NTRK1/2/3 fusions or Oncomine™ Comprehensive Assay (OCA) for detecting NTRK1/3 fusions.
Of this entire cohort, just one (0.04%) patient was identified with an ETV6-NTRK3 fusion. This patient was a 60-year-old man with a 35 pack-year smoking history, who was diagnosed with lung adenocarcinoma.
The investigators also evaluated NTRK1 gene fusion–induced imbalances in the expression of the 5’ and 3’ regions of the transcripts using the OFA sequencing data, but found no case of 5’/3’ expression imbalances of NTRK1 gene transcripts.
The sensitivity of NTRK fusions to Trk inhibition is being evaluated
ETV6-NTRK3 fusions have been detected in tumours having mesenchymal origin and have been identified in secretory carcinoma of the breast and in infantile fibrosarcoma and mesoblastic nephroma. A trial is currently underway that investigates whether ROS1, ALK, and NTRK fusions may be sensitive to Trk inhibition.
The STARTRK (NCT02568267) basket study is evaluating entrectinib (RXDX-101) in patients with solid tumours harbouring NTRK 1/2/3, ROS1, or ALK fusions.
Conclusions
The authors concluded that the frequency of NTRK fusions in Japanese patients with lung cancer is extremely rare (0.04%) compared with Caucasian patients.
Disclosure
Funding from Taiho, Chugai, Ono, Astra Zeneka, Bristol-Myers Squibb, MSD, Astellas, Eisai, DAIICHI SANKYO, Pfizer, Kyowa Hakko Kirin, Novartis, Takeda, Eli Lilly Japan, Merck Serono, Boehringer Ingelheim, and Amgen was reported.
Reference
415O – Nakamura A, et al. Prevalence of NTRK gene fusions in a large cohort of Japanese patients with lung cancer.