Induction chemotherapy with nab-paclitaxel plus gemcitabine or sequential FOLFIRINOX followed by surgical exploration induces high conversion rates in patients with locally advanced pancreatic cancer (LAPC) and conversion is associated with improved overall survival (OS), according to final results from the NEOLAP study that were presented at the ESMO Congress 2019 in Barcelona, Spain.
On behalf of the German Pancreatic Cancer Group (AIO-PAK), Volker Kunzmann of the Department of Internal Medicine II, Medical Oncology, University Hospital Wuerzburg in Wuerzburg and Comprehensive Cancer Centre Mainfranken, Germany presented the final results from the first prospective, randomised trial (NEOLAP; NCT02125136, EudraCT number: 2013-004796-12) comparing the efficacy and safety of either nab-paclitaxel plus gemcitabine or sequential fluorouracil, leucovorin, irinotecan, and oxaliplatin (sequential FOLFIRINOX) as induction chemotherapy in LAPC.
Approximately 30% of patients with pancreatic cancer present with locally advanced tumours and multiagent induction chemotherapy is recommended for this situation, but the optimal regimen is unknown. Conversion to resectable tumours in LAPC after chemotherapy and/or radiotherapy has been reported at variable frequencies in larger prospective multicentre trials, usually based on radiographic resectability criteria.
Professor Kunzmann and colleagues conducted an open-label, randomised, two-arm, phase II trial in 168 treatment-naive patients with histologically and/or cytologically confirmed non-resectable LAPC who were recruited from 33 German centres. The NEOLAP is the first prospective, randomised study to compare two different induction chemotherpy regimen (nab-paclitaxel/gemcitabine versus FOLFIRINOX-based) in that setting.
The patients received 2 cycles of nab-paclitaxel/gemcitabine induction; following these treatment cycles, 130 patients without progressive disease or unacceptable adverse events were randomly assigned 1:1 to receive either 2 additional cycles of nab-paclitaxel/gemcitabine (n=64) or 4 cycles of sequential unmodified sequential FOLFIRINOX (n=66).
Secondary resectability was assessed by surgical exploration in all patients achieving stable disease or a response after completion of induction chemotherapy. Complete macroscopic tumour resection (R0/R1 resection) served as the primary endpoint and secondary endpoints included safety, overall response rate, disease control rate (DCR), CA 19-9 response, pathological response, recurrence-free survival, progression-free survival and OS.
Following treatment with nab-paclitaxel/gemcitabine treatment and sequential FOLFIRINOX the DCR were 82.3% compared to 75.0% in the respective treatment arms (p = 0.38).
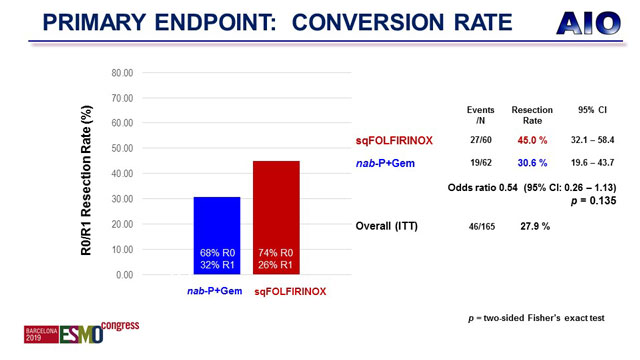
Surgical exploration was possible in equivalent numbers of patients receiving each treatment and was performed in 62.5% of patients in the nab-paclitaxel/gemcitabine group and 63.6% in the sequential FOLFIRINOX group.
The primary endpoint of the conversion rate was 30.6% with nab-paclitaxel/gemcitabine compared to 45.0% with sequential FOLFIRINOX, odds ratio (OR) 0.54; 95% confidence interval (CI), 0.26 -1.13 (p = 0.135).
Regarding the secondary efficacy endpoints, there was no difference between two groups. With a median follow-up of 13.8 months, the median OS was 17.2 months with nab-paclitaxel/gemcitabine compared to 22.5 months with sequential FOLFIRINOX, adjusted hazard ratio (HR) 0.73; 95% CI, 0.42 -1.28 (p = 0.268).
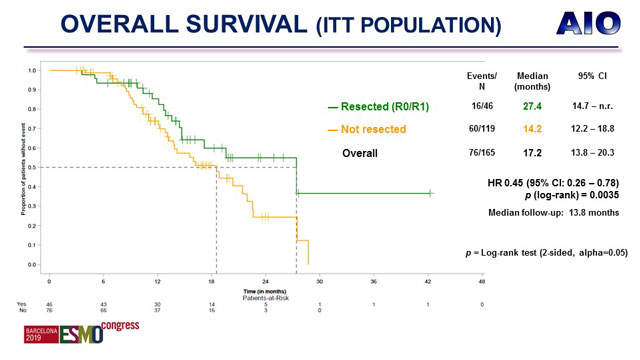
Patients achieving conversion had prolonged overall survival
Conversion associated with significantly improved OS in the 165 patients in the intent-to-treat (ITT) population; OS was 27.4 months in patients who converted compared to 14.2 months in those who did not (p = 0.0035).
The number of grade ≥3 adverse events were similar with the 2 treatments and were reported in 54.7% of patients in the nab-paclitaxel/gemcitabine group and in 53.0% of patients receiving sequential FOLFIRINOX.
Discussant points
Per Pfeiffer of the Department of Oncology, Odense University Hospital, Odense, Denmark who discussed the study findings said that is not in favour of randomised trials with 3 arms but a FFX arm might have given important information. More data on radiological evaluation is expected and until otherwise proven, laparotomy after conversion therapy is recommended. In terms of LAPC vs borderline resectable disease, exact stage must be reported. Participation in trials is recommended.
Conclusions
According to the authors, sequential FOLFIRINOX was not significantly superior to 4 month induction chemotherapy with nab-paclitaxel/gemcitabine in terms of conversion rate as the study primary endpoint, as well as in terms of secondary efficacy endpoints. Both induction chemotherapy regimens were tolerable and consistent with their known safety profiles. However, secondary tumour resection after obligatory surgical exploration was associated with significant survival benefit and it is highly recommendable in patients with LAPC.
Results of ongoing central radiographic review in terms of resectability status and response rate, as well as translational biomarker analyses from this study will be presented at future meetings.
Disclosure
Funding from Celgene was disclosed.
Reference
671O – Kunzmann V, Algül H, Goekkurt E, et al. Conversion rate in locally advanced pancreatic cancer (LAPC) after nab-Paclitaxel/Gemcitabine- or FOLFIRINOX-based induction chemotherapy (NEOLAP) - Final Results of a multicenter randomised Phase 2 AIO trial.