Maintenance therapy with olaparib following platinum-based chemotherapy provided a substantial benefit with regard to progression-free survival (PFS) among women with newly diagnosed advanced ovarian cancer and a BRCA1 and/or BRCA2 mutation (BRCAm), according to long-term SOLO1 findings presented at the ESMO Asia Virtual Congress 2020, held from 20 to 22 November 2020.
Michael Friedlander of the Medical Oncology Department, University of New South Wales Clinical School, Prince of Wales Hospital in Randwick, Australia presented updated findings on behalf of colleagues with 5 years of follow-up from the SOLO1 (NCT01844986; GOG-3004) study. He noted that, because of the high risk of relapse in patients with newly diagnosed advanced ovarian cancer, the 5-year survival rates are just 30–50%, which makes delaying recurrence, prolonging survival, and increasing the chance of cure the primary treatment goals.
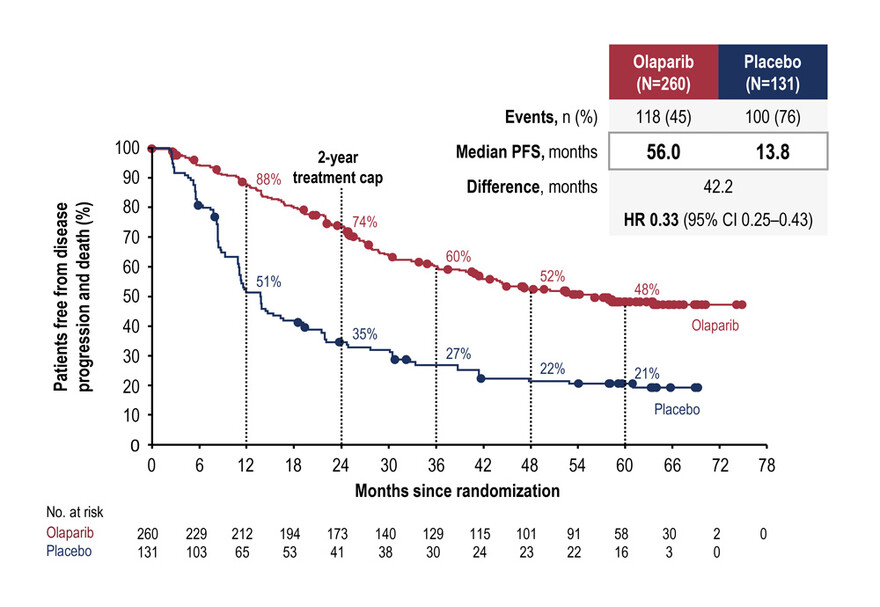
The SOLO1 study enrolled patients with ovarian cancer and a BRCAm who were in clinical response after first-line platinum-based chemotherapy. Following 2:1 randomisation, 260 patients received maintenance oral olaparib in 300 mg tablets twice daily and 131 patients received placebo for up to 2 years or until progression. Previously reported results from SOLO1 demonstrated a significant PFS benefit from maintenance olaparib; with median follow-up of 41 months, the median PFS was not reached (NR) with olaparib, compared with 13.8 months with placebo (hazard ratio [HR] 0.30; p < 0.001).1
The primary endpoint was investigator-assessed PFS per modified RECIST v1.1. For patients in complete response (CR) at baseline, recurrence-free survival (RFS) was defined post hoc as time from randomisation to disease recurrence characterised by new lesions by imaging, or death.
Olaparib maintenance reduced the risk of disease recurrence or death by 63% in patients with baseline complete response
The median treatment duration was 24.6 months with olaparib and 13.9 months with placebo; median follow-up was 4.8 and 5.0 years, respectively.
During follow-up, PFS events were observed in 45% of patients on olaparib versus 76% of patients on placebo. With olaparib maintenance, median PFS was 56.0 months, compared with 13.8 months with placebo (HR 0.33; 95% confidence interval [CI] 0.25–0.43). At 1, 2, 3, 4, and 5 years, 87.7%, 73.6%, 60.1%, 52.3%, and 48.3% of patients receiving olaparib maintence were progression-free, compared with 51.4%, 34.6%, 26.9%, 21.5%, and 20.5% of patients receiving placebo, respectively.
Among patients in CR at baseline, median RFS was NR in 189 patients receiving olaparib, compared with 15.3 months in 101 patients receiving placebo (HR 0.37; 95% CI 0.27–0.52), which tranlates to a 63% reduction in the risk of disease recurrence or death. In these patients, at 1, 2, 3, 4, and 5 years, 91.0%, 77.2%, 64.0%, 55.2%, and 51.9% of olaparib-treated patients were recurrence-free, compared with 58.0%, 39.0%, 28.9%, 23.0%, and 21.8% of patients receiving placebo, respectively.
Maintenance olaparib demonstrated a safety profile that was consistent with previous observations.
No new cases of myelodysplastic syndrome or acute myeloid leukaemia were reported, and the incidence of new primary malignancies remained balanced between arms at 3% with olaparib and 4% with placebo.
Conclusions
According to the investigators, patients with a BRCAm and newly diagnosed advanced ovarian cancer sustained the benefit provided by 2 years of maintenance olaparib beyond the end of treatment; after 5 years, almost half of the patients were progression-free, compared with 20% of patients receiving placebo.
In addition, over 50% of patients in CR after first-line platinum-based chemotherapy remained free from relapse 5 years later.
They noted that the 5-year follow-up in this study is the longest for any PARP inhibitor in this setting, and no new safety signals were observed.
This study was sponsored by AstraZeneca and is part of an alliance between AstraZeneca and Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA.
Citation
- Moore K, et al. N Engl J Med 2018;379:2495-2505. DOI: 10.1056/NEJMoa181085.
Reference
234O – Friedlander ML, Moore K, Colombo N et al. Maintenance olaparib for patients (pts) with newly diagnosed, advanced ovarian cancer (OC) and a BRCA mutation (BRCAm): 5-year (y) follow-up (f/u) from SOLO1. ESMO Asia Virtual Congress 2020 (20–22 November).